Description
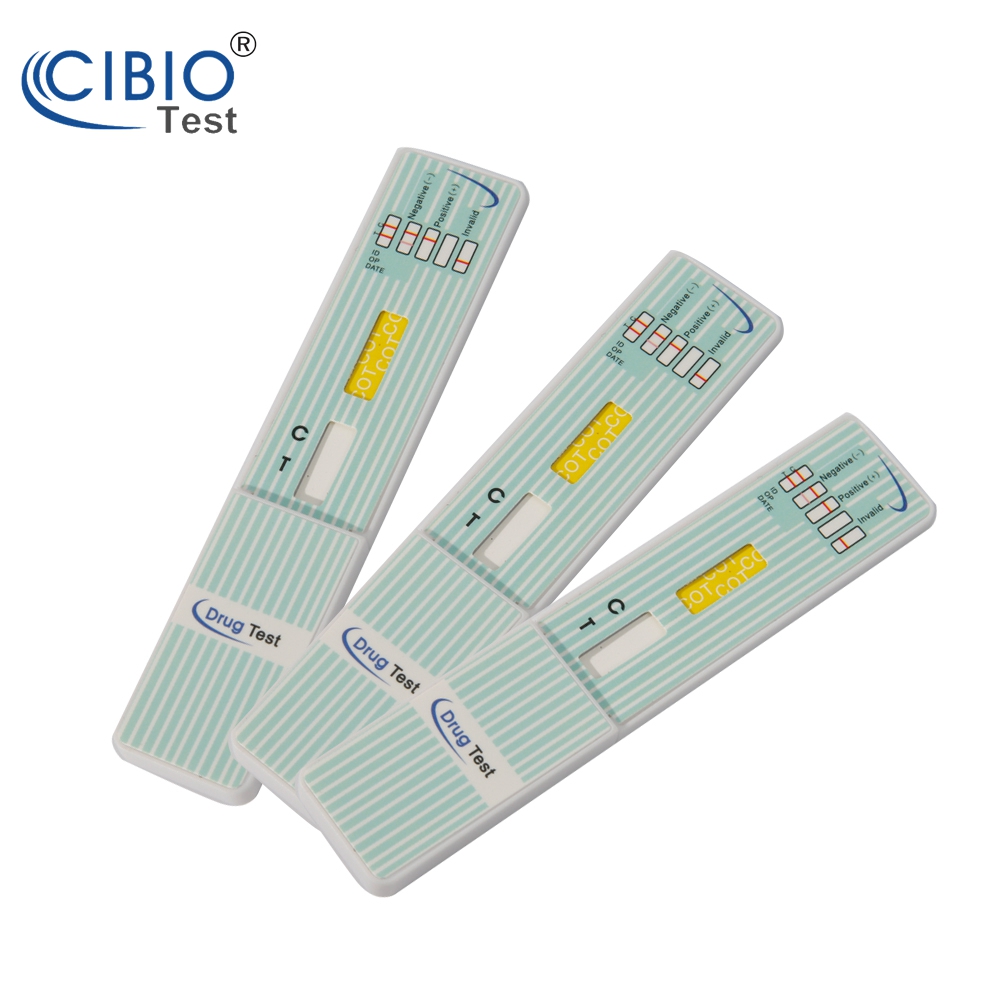
Single Panel Drug Test Dipcard
Rapid Single Drug Test kits is a rapid, screening test for the qualitative detection of single drug and drug metabolites in human urine at specified cut off levels.
For professional use only. For in vitro diagnostic use only.

| Product Name | Single Panel Drug Test Dipcard |
| Brand Name | CIBIO® or OEM |
| Format | Dip-card |
| Methodology | Colloidal Gold |
| Kit Contents | 1. Test Strip |
| 2. Desiccant | |
| 3. Instruction for use | |
| Assay Type | Qualitative Detection |
| Specimen | Urine |
| Specification | Dipcard size: 4.0 mm |
| Certificates | ISO 13485, FDA 510K, Clia Waived, CE |
| Accuracy | Over 99% |
| Storage Temperature | 4-30 degree centigrade |
| Shelf Life | 3 years ( 36 months ) |
| Delivery time | 15-30 days |
| OEM Services | Pouch, Box, Insert, private label |
| Loading Port | Guangzhou or Shenzhen |
Toxicology urine test list
| Product Description | Abbreviations | Cut-off Level | Qualification | ||
| 1 | Amphetamine | AMP | 1000 ng/mL | CE | FDA 510K, CLIA-waived |
| 2 | Barbiturates | BAR | 300 ng/mL | CE | FDA 510K, CLIA-waived |
| 3 | Buprenorphine | BUP | 10 ng/mL | CE | FDA 510K, CLIA-waived |
| 4 | Benzodiazepines | BZO | 300 ng/mL | CE | FDA 510K, CLIA-waived |
| 5 | Cocaine | COC | 300 ng/mL | CE | FDA 510K, CLIA-waived |
| 6 | Cotinine | COT | 200 ng/mL | CE | |
| 7 | Ethylglucuronide | ETG | 500 ng/mL | CE | |
| 8 | Fentanyl | FYL | 200 ng/mL | CE | |
| 9 | Fentanyl | FYL | 1 ng/mL | FDA 510K | |
| 10 | Synthetic Cannabinoid | K2 | 50 ng/mL | CE | |
| 11 | Ketamine | KET | 1000 ng/mL | CE | |
| 12 | 6-Acetylmorphine | 6-MAM | 10 ng/mL | CE | |
| 13 | Methylenedioxymethamphetamine – ecstasy | MDMA | 500 ng/mL | CE | FDA 510K, CLIA-waived |
| 14 | Methamphetamine | MET | 1000 ng/mL | CE | FDA 510K, CLIA-waived |
| 15 | Morphine | MOP | 300 / 2000 ng/mL | CE | FDA 510K, CLIA-waived |
| 16 | Methadone | MTD | 300 ng/mL | CE | FDA 510K, CLIA-waived |
| 17 | Opiate 2000 | OPI | 2000 ng/mL | CE | |
| 18 | Oxycodone | OXY | 100 ng/mL | CE | FDA 510K, CLIA-waived |
| 19 | Phencyclidine | PCP | 25 ng/mL | CE | FDA 510K, CLIA-waived |
| 20 | Propoxyphene | PPX | 300 ng/mL | CE | FDA 510K, CLIA-waived |
| 21 | Tri-cyclic Antidepressants | TCA | 1000 ng/mL | CE | FDA 510K, CLIA-waived |
| 22 | Marijuana | THC | 50 ng/mL | CE | FDA 510K, CLIA-waived |
| 23 | Marijuana | THC | 20 / 50ng/mL | FDA 510K, CLIA-waived | |
| 24 | Tramadol | TRA | 200 ng/mL | CE | |
| 25 | Xylazine | XYL | 1000 ng/mL | ||
| 26 | Methaqualone | MQL | 300 ng/mL | CE | |
| 27 | Carisoprodol | SOMA | 1000 ng/mL | ||
| 28 | Methcathinone | MCAT | 500 ng/mL | ||
| 29 | Lysergic Acid Diethylamide | LSD | 20 ng/mL | ||
| 30 | Kratom | KRA | 500 ng/mL | ||
| 31 | 3,4-Methylenedioxypyrovalerone | MDPV | 1000 ng/mL | ||
| 32 | Nitazene | NTZ | 100 ng/mL | ||
| 33 | Gabapentin | GAB | 1000 ng/mL | ||
| 34 | Tianeptine | TIA | 500 ng/mL | ||
| 35 | Medetomidine | MED | 1000 ng/mL | ||
Optional Adulterant Strips: CR – Creatinine, OX – Oxidants, pH – Acidic or Alkaline, S.G – Specific Gravity, NIT – Nitrite, GLU – Glutaraldehyde
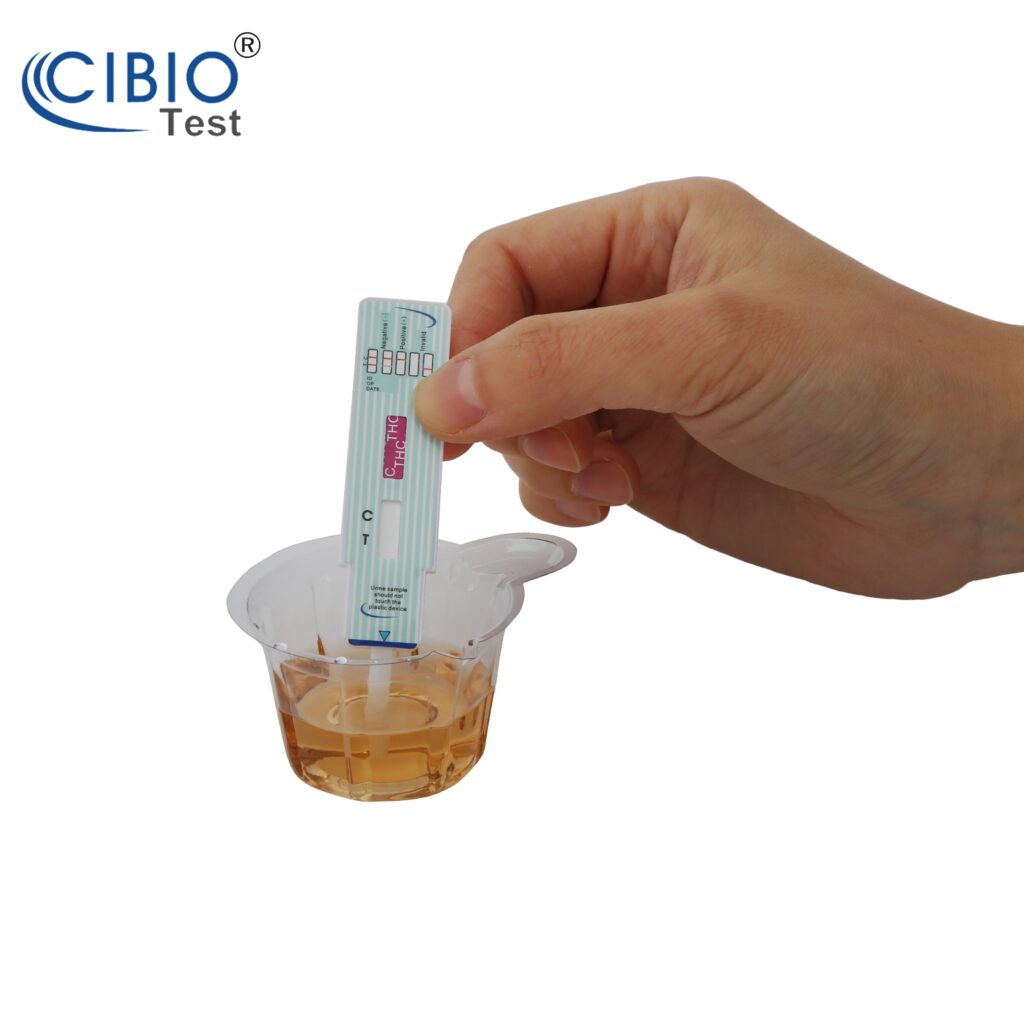
How to do the test?
Test must be at room temperature 59-86°F (15-30°C).
1. Open the sealed pouch by tearing along the notch. Remove the test dipcard from the pouch.
2. Immerse the dipcard into the urine with the arrow pointing towards the urine. Take the dipcard out after 10 seconds.
IMPORTANT:Do not allow the urine level to exceed the MAX (marker line), otherwise the test will not perform correctly.
3. Lay the test strip on a clean, dry, non-absorbent surface.
4. Read the results at 5 minutes. The drug test results remain stable for up to thirty minutes.
See the illustration.
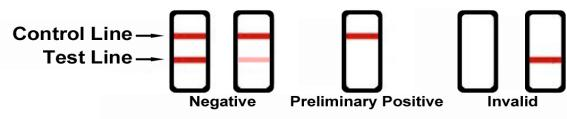
Reading the results
Preliminary positive (+)
A rose-pink band is visible in the control region, but no colored band appears in the test region. This indicates a preliminary positive result for this specific drug. Preliminary positive results must be sent to a lab for a more reliable test.
Negative (-)
A rose-pink band is visible in the control region and the test region. This indicates that the drug is not present in the sample or is at a level that cannot be detected by this test.
Invalid
If a color band is not visible in the control region, or a color band is only visible in the test region, the test is invalid. You should repeat the test with a new test Strip. If the test still shows an invalid result, please contact us at 1-864-538-8007. When calling, be sure to provide the lot number for the test.
Note: There is no meaning to the colored line intensity or width. Any visible line is considered to be a line.
Certain lines may appear lighter or thinner than other lines.
ANY COLORED LINE VISIBLE IN THE TEST “T” REGION, NO MATTER HOW DARK OR FAINT, SHOULD BE INTERPRETED AS A NEGATIVE RESULT AS LONG AS THE CONTROL LINE IS ALSO PRESENT.
IMPORTANT:
The test Strip provides only a preliminary test result. All “preliminary positive” samples must be sent to a laboratory for a more reliable test.
Q&A
Qus: How do rapid drug tests work?
Rapid drug tests detect the presence of drugs and drug metabolites in human urine at specified cut off levels in minutes. When a detection strip is wetted with urine specimen, the specimen reacts with the reagents and antigens on the strip and produces a color reading so you can interpret a positive or negative result.
Qus: What is the difference between a Dip Test strip and a Cassette Test?
The Dip strip is dipped into a urine specimen to get results.
A cassette is a convenient way that the specimen can drop into sample well.
Qus: What is the shelf life of your drug testing products?
The rapid drug test products have a shelf life of 36 months from manufacturing date. The expiration date is on every individual pack.
Qus: The drug line is lighter than the control line. Does this mean some drug is present?
No. Any line next to the word Drug or the drug abbreviation (depending on the test you have purchased), no matter how dark or light, is considered a Negative Result and no further testing is required.
Qus: Why is Urine Testing the Most Frequently Used Method for Drug Testing?
Businesses choose to drug test their workers and new job applicants to reduce the risk of workplace drug use and relevant hazards. While all drug test types, like hair follicle, blood, and saliva, have their benefits, urine drug testing is the most flexible and customizable.






Reviews
There are no reviews yet.